Why Choose Left When You Can Be Right?
Introducing Azogen 5-0-0, our crystal-clear organic liquid ammoniacal fertilizer.
*The image is for illustrative purposes only.


A Unique Natural Product
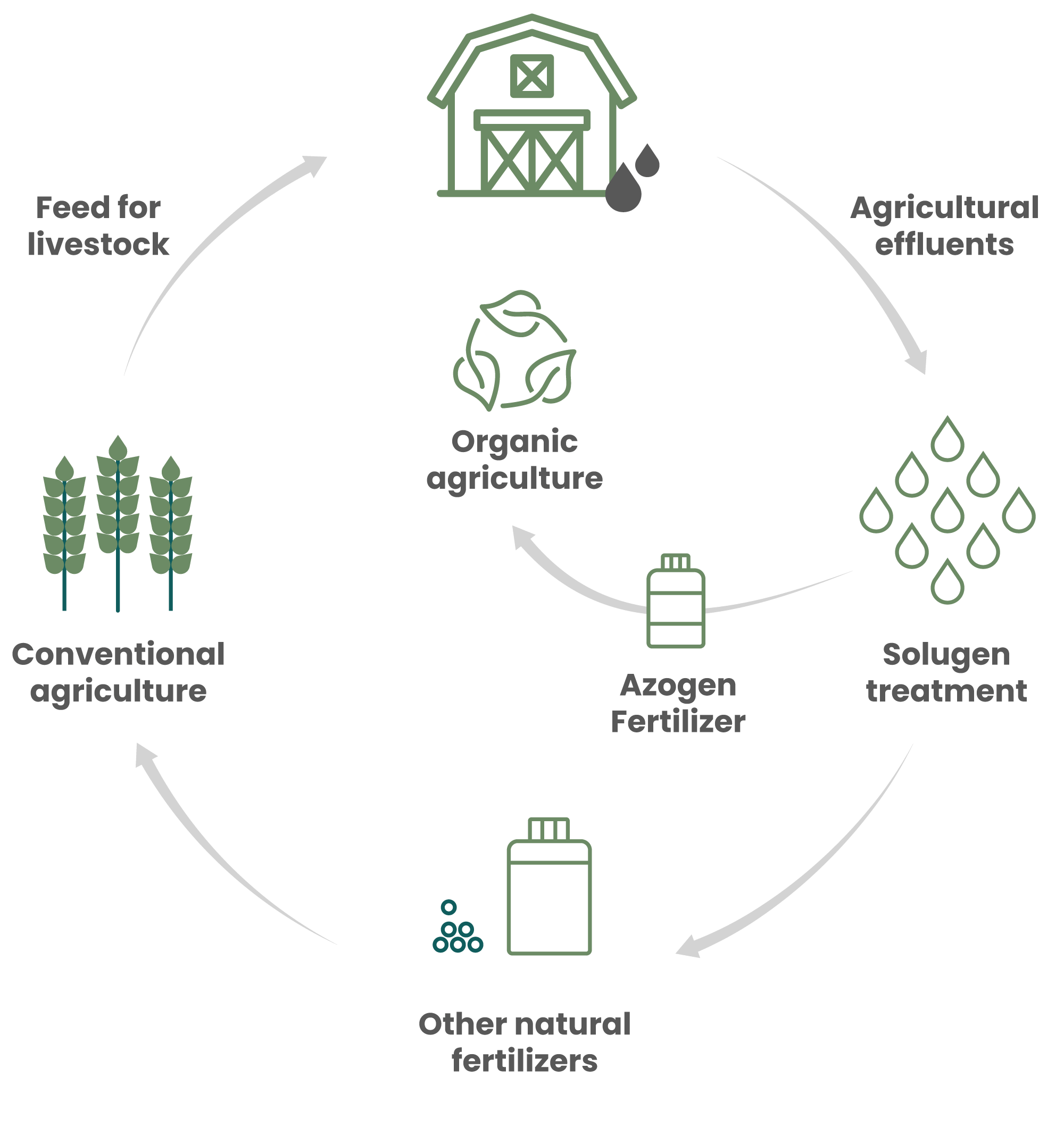
OMRI listed Azogen 5-0-0, an organic liquid nitrogen fertilizer, is an entirely natural product derived from a unique process developed by Solugen Global. This process captures and recovers ammonia emissions from livestock farming, transforming them into a liquid nitrogen fertilizer. The sale of this product supports the financing of new agricultural effluent treatment plants, contributing to the reduction of greenhouse gases generated by the livestock industry during effluent storage and spreading, and minimizing their impact on the environment.
Inspired by the Circular Economy
"At Solugen Global we understand the importance and value of organic agriculture. By creating a circular economy based model, we help make the world a better place by protecting ecosystems."
— Andre Beaulieu Blanchette, CEO
-
Production Capacity
-
Benefits
-
Innovation
-
An Endless Production Capacity
With our production plan in effect since December 2023, we will produce significant quantities of Azogen 5-0-0 annually. Additionally, we are already planning to increase production over the next 2 years. -
The environmental benefits of Solugen Global’s process
By treating 10,000 m3 of liquid agricultural effluent per year, which is representative of a medium-sized farm in North America, Solugen Global's process reduces the number of times that liquid agricultural effluent is transported to cultivated land by 425. Solugen Global's technology contributes to the reduction of greenhouse gases. The treatment of 10,000 m3 of liquid agricultural effluent results in a reduction of 572 tons of CO2 equivalent. Solugen Global's process catches and recovers ammonia from agricultural effluents, thereby preventing it from volatilizing and becoming an atmospheric pollutant. Water is a global issue. Solugen Global's technology recovers 75% of the treated volume as pure, reusable water. By concentrating most of the phosphorus in a biosolid residue extracted from Solugen Global's treatment, phosphorus management is optimized, greatly reducing the risk of eutrophication in waterways. Our plant can treat huge volumes of agricultural effluent per year. -
An innovation developed by Solugen Global
Our mission is to produce and market organic fertilizers according to the logic of circular economy by treating agricultural effluents with an innovative process that reduces the environmental footprint of livestock breeding. Our vision is to become the world-renowned Canadian leader in the sustainable production of organic fertilizers by March 31, 2027. Our values are based on three axes: professionalism, adaptability, and team spirit.
Optimal Application Recommendations
-
Fertigation
-
Berries
50-150 ppm N
-
Vegetables
50-150 ppm N
-
-
In Furrow
-
Fields crops
10 to 35 gal/acre
-
Performance of any fertilizer product may be influenced by local climatic conditions, cropping patterns, and soil/substrate types. The provided general information is intended as a guideline. We strongly recommend seeking prior advice on dosage and application frequency tailored to the specific crop and growing conditions.
Technical Sheet

Azogen Fertilizer: Fertile in Benefits
-
1. Single-Element Formulation
Avoid over-fertilization risks with Azogen's single-element formulation. Ensure a suitable nitrogen supply for healthy plant growth.
-
2. Liquid Ease of Use
Azogen 5-0-0's liquid formulation offers versatile application methods, including field irrigation, soil injection, drip irrigation, and hydroponic solutions. Always dilute Azogen 5-0-0 in water.
-
3. Highly Versatile
Azogen 5-0-0 is perfect for crops, flowering plants, berries, turf, and lawns, showcasing its exceptional versatility.
-
4. Quality Nitrogen
Comprising mainly ammoniacal nitrogen, Azogen 5-0-0 ensures rapid absorption by plants.
-
5. Environmental Contribution
Choose Azogen 5-0-0 for a substantial reduction in greenhouse gases, soil protection, and waterway preservation, making a positive impact on the environment.
Packaging Options
-
Truckload (tanker) 5500 US gallons
Please contact us for a quote
-
265 US gallon tote
Please contact us for a quote
-
55 US gallon drum (4 x pallet)
Please contact us for a quote
-
2.5 US gallon
Please contact us for a quote
To inquire about alternative packaging options, please feel free to contact us.
Contact usContact Us
If you’re looking for an organic fertilizer manufacturer who can expand production capacity to meet your customers needs, feel free to contact us.
Contact Details
By email
If you are a grower: sales@solugenglobal.com
If you are a distributor: distributors@solugenglobal.com
General inquiries: info@solugenglobal.com
By phone
1-844-729-6436
(1-844-7AZOGEN)

